 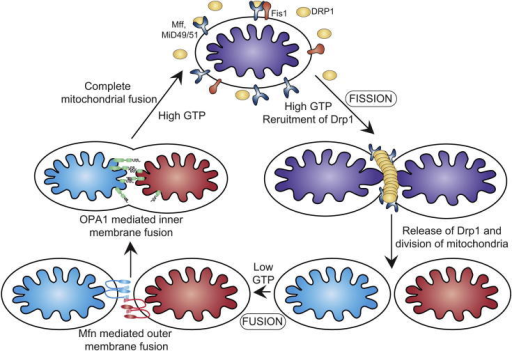
Phosphomimetic substitution at Ser637Asp was also shown to block mitochondrial fission ( Chang & Blackstone, 2007). An independent study ( Chang & Blackstone, 2007) corroborates these findings by showing that PKA-dependent phosphorylation of Drp1 within the GED domain at Ser 637 blocks Drp1 GTPase activity. In this issue, Cribbs & Strack have addressed some of these important questions and provide a mechanistic link for second messenger regulation of Drp1 GTPase activity and apoptosis signalling.įirst, Cribbs & Strack show that PKA-mediated-phosphorylation of Drp1 at Ser 656 attenuates the GTPase activity of Drp1 and promotes cell survival, suggesting that cAMP might mediate survival partly through the inhibition of Drp1 ( Fig 1). In addition, although Drp1 GTPase can be regulated by ubiquitination and sumoylation ( Nakamura et al, 2006 Wasiak et al, 2007), there is little insight as to how Drp1 activation might be regulated during apoptosis signalling. Although gain- and loss-of-function studies of Drp1 correlate mitochondrial fission with apoptosis ( Frank et al, 2001 Germain et al, 2005), there is no evidence to show that Drp1 alone, or mitochondrial fission by itself, can induce apoptosis. The energy generated by GTP hydrolysis is believed to provide the mechanical force required to execute fission ( Ingerman et al, 2005). These studies provide a new mechanistic insight into the link between the mitochondrial fission machinery and cell death signalling.ĭrp1 is recruited to the mitochondrial surface at potential fission sites ( Ingerman et al, 2005 Okamoto & Shaw, 2005). Cyclic-AMP-dependent protein kinase (PKA)-mediated phosphorylation of Drp1 at Ser 656 induces mitochondrial elongation and resistance to apoptotic stimuli, whereas dephosphorylation of Ser 656 by calcineurin promotes mitochondrial fragmentation and increases cell vulnerability to apoptosis. In this issue of EMBO reports, Cribbs & Strack identify a new mechanism by which second messengers-cAMP and calcium-modulate mitochondrial shape and function through the regulation of Drp1 phosphorylation. Although the list of genes for mitochondrial morphogenesis is rapidly increasing, dynamin-related protein 1 (Drp1)-a cytosolic dynamin GTPase-was among the first fission proteins to be discovered however, the mechanism by which Drp1 function is regulated is poorly understood. Perturbation of the steady state between these opposing processes has been directly implicated in several human disorders ( Chan, 2006). Mitochondrial morphology, size and position within cells are maintained through a balance of fission and fusion events. Recent research has opened new avenues for the evaluation of mitochondrial function for example, mitochondria are no longer perceived as thread-like static entities within the cytosol, but instead are viewed as highly dynamic organelles that can change in shape and size, and are transported to strategic locations within the cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
